

Background
Pharmacovigilance industry is indeed on its hike and though we have global regulations for a common platform to report for product safety, each Company has its own style of working and to adapt to the same is indeed a challenge. Sarjen’s as a solution provider made sure to turn this challenge to an opportunity when different clients concerns are addressed and operations are embedded in solution to suit best industry practices.
One of the clients wanted to have a user defined workflow for routing cases from data entry user till submission user. Client wanted to ensure the medical review or QC review done is recorded in PV system and was willing to capture/save remarks of each user workflow as and when the ICSR/case was sent to subsequent workflows.
There was another client who wanted a full-proof system to avoid any accidental conflict of interest. Client insisted on having a system where same user would never be able to review/QC the case when case was created by him as a matter of compliance!
Analysis
Case routing process was analyzed for different operational practices in various set ups where product safety is handled by a team in large, medium or small scale setting.
Case routing was given a thought also from a global perspective where an authorized user could manage the case routing to each workflow by assigning a case to specific set of users when case is being sent!
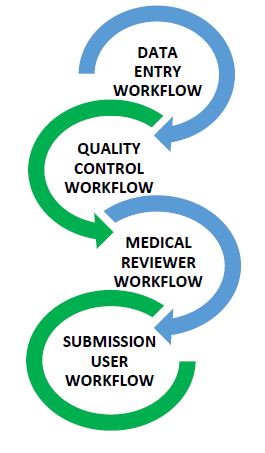
Solution
After understanding the process undertaken in different pharmacovigilance settings, it was finally decided to opt for two strategies:
- To allow user-based allocation of a case
- Follow similar process when case was routed back to previous workflows for any reasons or corrections
- To give flexibility of pre-defined set workflow where case can be passed in further stages to specific user as allocated by PV manager/authorized user.
Paybacks
- A robust system to meet the standard yet different practices
- User-defined case routing right from ICSR creation
- Audit trail for user delegation maintained
- Traceable remarks/comments for case sent to next or previous workflow
- Justifies requirements of larger as well as smaller PV settings to strictly follow user-assigned workflows
- Allows forward as well as reverse routing for case life cycle management
- Delegation can be done by team lead/managers in case of over load
- Justifies the user role in PV operations in audit and otherwise

